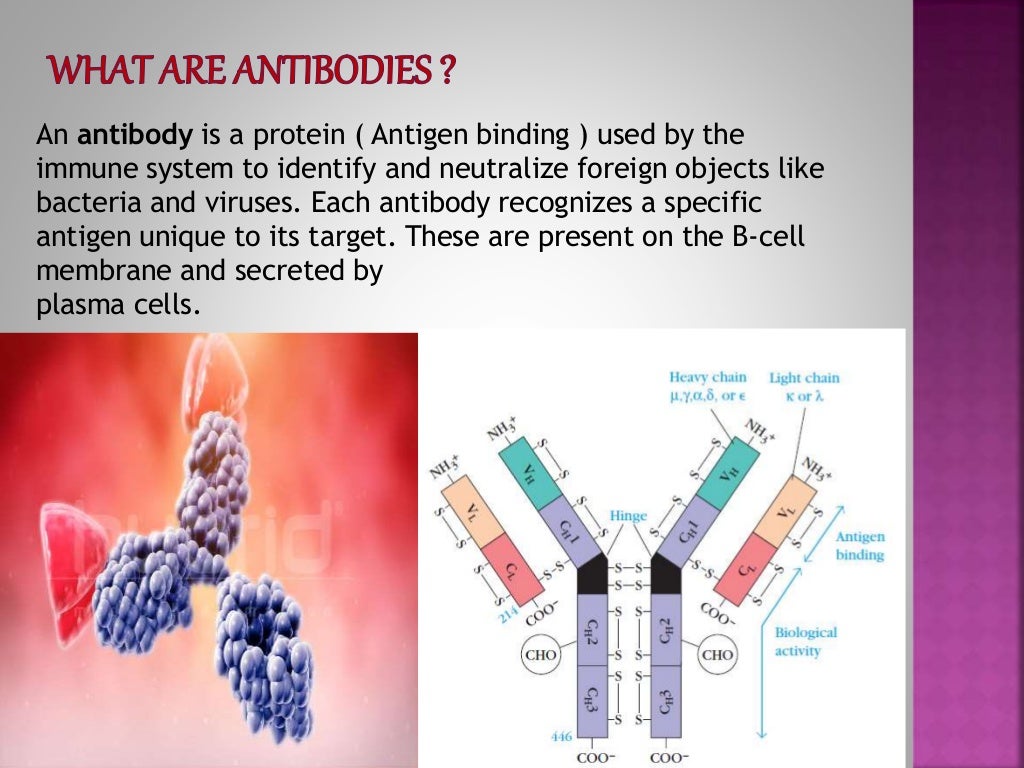
What do you know about monoclonal antibody therapy?
Sep 11, 2014 · 2. Generation of monoclonal antibodies using the hybridoma technique. Monoclonal antibodies are monovalent antibodies which bind to the same epitope and are produced from a single B-lymphocyte clone . They were first generated in mice in 1975 using a hybridoma technique . The generation of hybridomas involves immunising a certain species …
How effective is the monoclonal treatment?
Beginning on May 6, 2021, Medicare established separate coding and payment for administering COVID-19 monoclonal antibody products through infusion in a patient’s home or residence.
Who pays for monoclonal treatment?
Sep 28, 2021 · Publications were identified primarily through a search including the following Key search terms: casirivimab and imdevimab OR bamlanivimab and etesevimab OR mAb OR antibody treatment OR monoclonal antibody AND COVID-19 OR SARS-CoV-2 from January 01, 2020, to September 15, 2021.
What are the dangers of monoclonal antibodies?
Jan 06, 2022 · Individuals qualify for monoclonal antibody treatment if: they have tested positive for COVID-19, and it has been 10 days or less since symptoms first started, and they have other health conditions that put them at higher risk. Monoclonal antibody treatment is most effective when given early—and the sooner it is given, the better.

How many types of monoclonal antibody COVID-19 treatments are there in the US?
In the United States, there are three anti-SARS-CoV-2 monoclonal antibody treatments with FDA Emergency Use Authorization (EUA) for the treatment of COVID-19: bamlanivimab plus etesevimab, casirivimab plus imdevimab,, and sotrovimab.
How do monoclonal antibodies work against COVID-19?
Monoclonal antibodies for COVID-19 may block the virus that causes COVID-19 from attaching to human cells, making it more difficult for the virus to reproduce and cause harm. Monoclonal antibodies may also neutralize a virus.Mar 31, 2022
What is the first drug that was approved by the FDA to treat COVID-19?
Remdesivir is the first drug approved by the FDA for treatment of hospitalized COVID patients over the age of 12.Jan 25, 2022
What does monoclonal antibody treatment mean for COVID-19?
Monoclonal antibodies are manmade versions of the antibodies that our bodies naturally make to fight invaders, such as the SARS-CoV-2 virus.Apr 1, 2022
What is a monoclonal antibody?
Monoclonal antibodies are laboratory-produced molecules that act as substitute antibodies that can restore, enhance or mimic the immune system's attack on cells.Mar 31, 2022
Can I get the COVID-19 vaccine if I was treated with monoclonal antibodies or convalescent plasma?
If you were treated for COVID-19 symptoms with monoclonal antibodies or convalescent plasma, you should wait 90 days before getting a COVID-19 vaccine.
Is there a drug treatment for COVID-19?
The U.S. Food and Drug Administration has approved one drug treatment for COVID-19 and has authorized others for emergency use during this public health emergency. In addition, many more therapies are being tested in clinical trials to evaluate whether they are safe and effective in combating COVID-19.Jan 27, 2022
What is Remdesivir?
Remdesivir is in a class of medications called antivirals. It works by stopping the virus from spreading in the body.
Is Remdesivir approved for treatment of COVID-19?
Remdesivir is approved by the Food and Drug Administration (FDA) for the treatment of COVID-19 in hospitalized adult and pediatric patients (aged ≥12 years and weighing ≥40 kg).
Should you still get the COVID-19 vaccine if you were treated with monoclonal antibodies?
If you were treated for COVID-19 with monoclonal antibodies or convalescent plasma, there is no need to delay getting a COVID-19 vaccine.Feb 17, 2022
What is the difference between monoclonal antibodies and the COVID-19 vaccine?
COVID-19 vaccines help stimulate and prepare a person's immune system to respond if they are exposed to the virus. However, monoclonal antibodies boost the immune system only after a person is already sick, speeding up their immune response to prevent COVID-19 from getting worse.Nov 8, 2021
Do I need the COVID-19 vaccine if I still have antibodies?
Yes, the COVID-19 vaccines are recommended, even if you had COVID-19.Nov 23, 2021
COVID-19 VEKLURYTM (remdesivir)
Following the recent statement from the National Institutes of Health (NIH) COVID-19 Treatment Guidelines Panel about therapies for the COVID-19 Omicron variant, CMS created HCPCS code J0248 for VEKLURY™ (remdesivir) antiviral medication when administered in an outpatient setting.
COVID-19 Monoclonal Antibody Products
The FDA authorized the following investigational monoclonal antibody product under EUA for pre-exposure prophylaxis of COVID-19:
Important Update about Viral Variants
On April 16, 2021, the FDA revoked the EUA for bamlanivimab, when administered alone , due to a sustained increase in COVID-19 viral variants in the U.S. that are resistant to the solo product.
Medicare Coverage for COVID-19 Monoclonal Antibody Products
During the COVID-19 public health emergency (PHE), Medicare will cover and pay for these infusions (when furnished consistent with their respective EUAs) the same way it covers and pays for COVID-19 vaccines.
Coding for the Administration of COVID-19 Monoclonal Antibody Products
CMS identified specific code (s) for each COVID-19 monoclonal antibody product and specific administration code (s) for Medicare payment:
Medicare Payment for Administering COVID-19 Monoclonal Antibody Products
To ensure immediate access during the COVID-19 PHE, Medicare covers and pays for these infusions and injections in accordance with Section 3713 of the Coronavirus Aid, Relief, and Economic Security Act (CARES Act) .
Billing for Administering COVID-19 Monoclonal Antibody Products
Health care providers can bill on a single claim for administering COVID-19 monoclonal antibody products, or submit claims on a roster bill.
What are monoclonal antibodies?
Our bodies naturally make antibodies to fight infections. However, if you haven’t received the COVID-19 vaccine or had a previous COVID-19 infection, your body will not have antibodies designed to recognize a new virus like SARS-CoV-2.
How does monoclonal antibody therapy help?
Monoclonal antibody therapy is a way of treating COVID-19 for people who have tested positive, have had mild symptoms for seven days or less, and are at high risk for developing more serious symptoms.
Who is eligible for monoclonal antibody therapy?
Given that COVID-19 vaccination provides strong protection against severe disease and need for hospitalization, monoclonal antibody therapy is an option for certain high-risk patients with COVID-19.
What is monoclonal antibody infusion?
The center has locations in Barron and Eau Claire. "A monoclonal antibody infusion is meant to boost your own body's immune system. These man-made antibodies are meant to mimic antibodies your immune system begins to make after being exposed to COVID-19," says Lori Arndt, a physician assistant in Infectious Diseases at Mayo Clinic Health System in ...
Why did the Wachsmuths get monoclonal antibodies?
The Wachsmuths qualified for the monoclonal antibodies due to age and other chronic health conditions that increased their chances of developing severe disease or requiring hospitalization. The day after their positive COVID-19 tests, Bob and Joyce received monoclonal antibody infusions at the same time in the same room at the clinic.
How long does it take for Bob to feel better after a blood test?
After the antibody infusion, Bob's symptoms continued to improve. Within several hours, Joyce began to feel much better, with no fever, chills or body aches. Lori says that their experience is consistent with other patients. "Most patients report improvement of symptoms with 24 to 48 hours after infusion," she says.
When will the Mayo Clinic open?
The Mayo Clinic COVID-19 Infusion Center opened in November 2020. The center has locations in Barron and Eau Claire.
Can family members receive infusions together?
Lori says it is not uncommon to have family members receive infusion treatments together. "We have had several family members come in together to receive treatment. It's a wonderful service to offer patients who may be feeling anxious or apprehensive about receiving treatment," says Lori.