
Monoclonal antibodies
| Products | Sotrovimab | Bebtelovimab | EVUSHELD (Tixagevimab/ Cilgavimab) |
| Authorized use (s) | Treatment of mild to moderate symptoms | Treatment of mild to moderate symptoms. | Prevention: before exposure |
| Age eligibility | Ages 12 years and older | Ages 12 years and older | Ages 12 years and older |
| Weight eligibility | 88 pounds or more | 88 pounds or more | 88 pounds or more |
| Other criteria for treatment | Test positive for SARS-CoV-2 Be within 1 ... | Test positive for SARS-CoV-2 Be within 7 ... | Not applicable |
Are there side effects of monoclonal antibody treatment?
Apr 07, 2022 · Follow All monoclonal antibody (mAb) therapies are in limited supply, and not everyone will be eligible for treatment. A prescription from a healthcare provider is required to receive any mAb therapy. Variants, like Omicron, may …
How effective is the monoclonal treatment?
Jan 06, 2022 · These antibodies are given to people directly through an intravenous (IV) infusion. How does monoclonal antibody therapy help? Monoclonal antibody therapy is a way of treating COVID-19 for people who have tested positive, have had mild symptoms for seven days or less, and are at high risk for developing more serious symptoms.
What are the dangers of monoclonal antibodies?
To receive a mAb you should be referred for treatment by your healthcare provider and directed to available infusion locations. If you do not have a healthcare provider, call the Combat COVID Monoclonal Antibodies Call Center at 1-877-332-6585 to find out who to talk with about your symptoms and treatment. DOH 825-031 March 2022
Can monoclonal antibodies kill you?
Dec 28, 2021 · All of the monoclonal antibodies we give are injections, mostly intravenous (IV) infusions. This means the drug goes directly into your blood through a small needle inserted into a vein in your arm. To receive a monoclonal antibody infusion, you must have tested positive for COVID-19 and meet at least one of the following requirements:
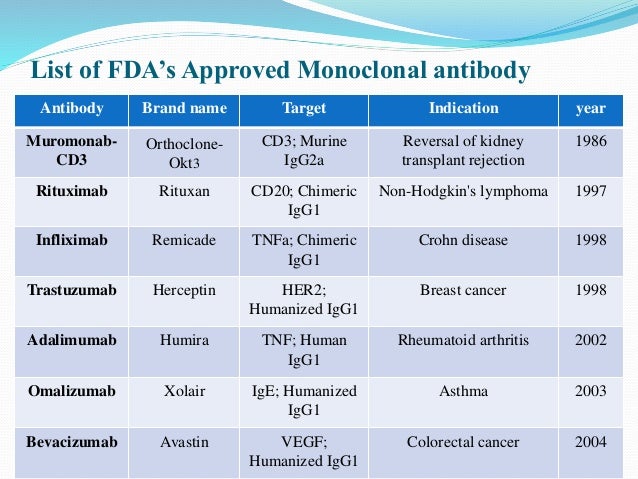
How do monoclonal antibodies work against COVID-19?
Monoclonal antibodies for COVID-19 may block the virus that causes COVID-19 from attaching to human cells, making it more difficult for the virus to reproduce and cause harm. Monoclonal antibodies may also neutralize a virus.Mar 31, 2022
How many types of monoclonal antibody COVID-19 treatments are there in the US?
In the United States, there are three anti-SARS-CoV-2 monoclonal antibody treatments with FDA Emergency Use Authorization (EUA) for the treatment of COVID-19: bamlanivimab plus etesevimab, casirivimab plus imdevimab,, and sotrovimab.
What is a monoclonal antibody?
Monoclonal antibodies are laboratory-produced molecules that act as substitute antibodies that can restore, enhance or mimic the immune system's attack on cells.Mar 31, 2022
Can I get the COVID-19 vaccine if I was treated with monoclonal antibodies or convalescent plasma?
If you were treated for COVID-19 symptoms with monoclonal antibodies or convalescent plasma, you should wait 90 days before getting a COVID-19 vaccine.
What is the first drug that was approved by the FDA to treat COVID-19?
Remdesivir is the first drug approved by the FDA for treatment of hospitalized COVID patients over the age of 12.Jan 25, 2022
Which drug is approved by FDA to treat COVID-19?
Veklury (Remdesivir) is an antiviral drug approved for use in adults and pediatric patients [12 years of age and older and weighing at least 40 kilograms (about 88 pounds)] for the treatment of COVID-19 requiring hospitalization.Mar 31, 2022
What is the difference between monoclonal antibodies and the COVID-19 vaccine?
COVID-19 vaccines help stimulate and prepare a person's immune system to respond if they are exposed to the virus. However, monoclonal antibodies boost the immune system only after a person is already sick, speeding up their immune response to prevent COVID-19 from getting worse.Nov 8, 2021
Should you still get the COVID-19 vaccine if you were treated with monoclonal antibodies?
If you were treated for COVID-19 with monoclonal antibodies or convalescent plasma, there is no need to delay getting a COVID-19 vaccine.Feb 17, 2022
Is there an antibody cocktail for COVID-19?
The treatment, bamlanivimab and etesevimab administered together, was granted FDA emergency use authorization in February. Eli Lilly and the FDA stipulated that the antibody cocktail is authorized as a COVID-19 prophylaxis only for individuals who have been exposed to the virus.Sep 16, 2021
What medication is not recommended before vaccinations for COVID-19?
It is not recommended you take over-the-counter medicine – such as ibuprofen, aspirin, or acetaminophen – before vaccination for the purpose of trying to prevent vaccine-related side effects. It is not known how these medications might affect how well the vaccine works.
Who should not take the Pfizer-BioNTech COVID-19 vaccine?
If you have had a severe allergic reaction to any ingredient in the Pfizer-BioNTech COVID-19 vaccine (such as polyethylene glycol), you should not get this vaccine. If you had a severe allergic reaction after getting a dose of the Pfizer-BioNTech COVID-19 vaccine, you should not get another dose of an mRNA vaccine.
What are the contraindications to the COVID-19 vaccine?
Contraindications to COVID-19 vaccination include: Severe allergic reaction (e.g., anaphylaxis) after a previous dose or to a component of the COVID-19 vaccine. Known diagnosed allergy to a component of the COVID-19 vaccine (see Appendix C for a list of vaccine components).
What is the FDA approved monoclonal antibody?
Department of Health and Human Services (HHS), two different monoclonal antibody treatments have been authorized for emergency use by the FDA. One is bamlanivimab, and the other is a mixture of casirivimab and imdevimab. Over 600,000 such treatments have been shipped to U.S. health care facilities.
Who is the chief science officer for the Biden administration?
Dr. David Kessler , chief science officer for the Biden administration's COVID response, has now said "it is very important" that Americans enquire about monoclonal antibodies if they test positive for coronavirus and fall into high-risk categories.
Is monoclonal antibody free?
The federal government is currently distributing monoclonal antibodies for free, but some health care providers may be charging patients for administering the treatment. These costs may be covered if the patient has insurance. If not, it is advised to ask the treatment facility if there will be any charges.
