How expensive are monoclonal antibodies?
Sep 24, 2021 · The monoclonal drug is expensive but the federal government is covering the cost. “The drug itself is provided free to the sites. That is significant because the drug normally costs between $3,000 to $5,000 a dose,” Dr. Michael Saag, UAB Infectious Diseases, said.
How effective is the monoclonal treatment?
Because the federal government has purchased a supply of certain monoclonal antibody treatments, there is no cost to the patient for the monoclonal antibody products themselves; however, there may be costs incurred from administration of the product. Checking insurance coverage is advised.
Which monoclonal antibody is best?
Aug 25, 2021 · The cost of GSK’s and Vir’s monoclonal antibody costs about $2,100 per infusion. This is covered by a combination of government payments, reimbursements, and GSK’s copay program, reports USA ...
Will insurance cover monoclonal antibody?
Results: The average annual price of a mAb was $96,731, exceeding $100,000 for 34 mAb-indication combinations. Oncology and hematology mAbs represented 40% of the mAb-indication combinations approved, yet they accounted for more than 85% of those priced $100,000 or higher.
Frequently Asked Questions
The following frequently asked questions will prepare providers for common questions about monoclonal antibody treatment cost and coverage in the outpatient setting. Other options are available for inpatient settings. 1
Additional Resources
Additional CMS guidance on coding, billing, payment allowances, and effective dates for COVID-19 Monoclonal Antibodies and their Administration During the Public Health Emergency:
What are monoclonal antibodies used for?
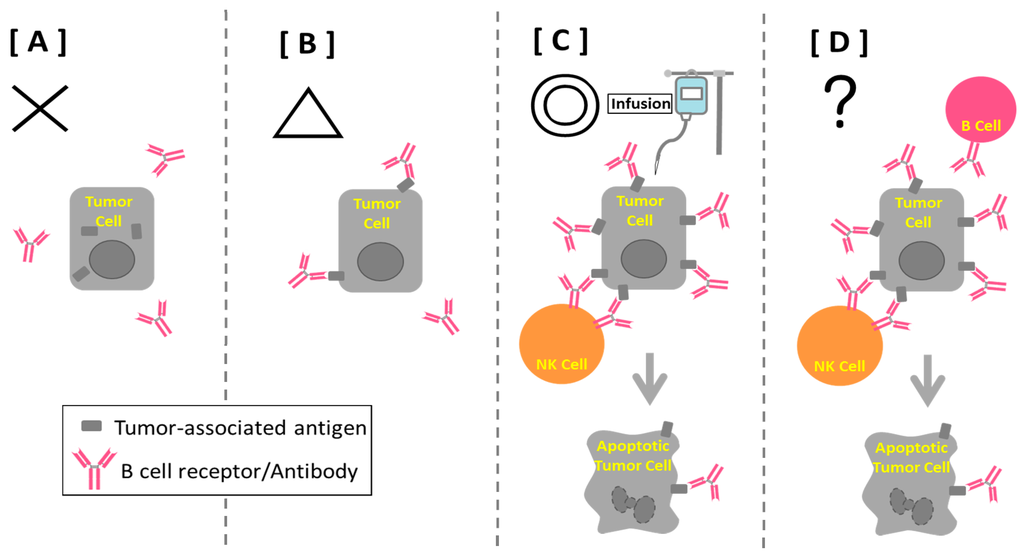
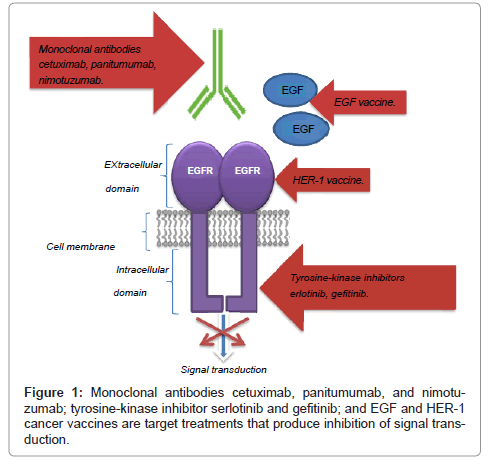
Monoclonal antibodies are used to treat many diseases, including cancer; autoimmune diseases like rheumatoid arthritis or Crohn's disease; and respiratory syncytial virus in children. They work by replacing or substituting antibodies in the body's immune system that specifically target certain antigens ...
What is the drug class for eculizumab?
Drug class: Monoclonal Antibodies. ECULIZUMAB is a monoclonal antibody. It is used to treat a rare kind of anemia called paroxysmal nocturnal hemoglobinuria or PNH. It may help prevent the loss of blood in patients with PNH.
What is palivizumab used for?
PALIVIZUMAB is an antibody. It is used in infants and children to prevent severe cases of respiratory syncytial virus (RSV) infection. Children treated with this medicine may still get RSV but will not get as sick as if they were not treated at all.
Is Casirivimab an antibody?
Drug class: Monoclonal Antibodies. Casirivimab is an investigational monoclonal antibody therapy authorized for emergency use by the FDA to treat COVID-19. It must be administered together with imdevimab. It is not yet FDA approved, and the safety and effectiveness of this therapy is still being evaluated.
Is Dupixent a generic drug?
Dupixent (dupilumab) is an expensive drug used to treat certain patients with eczema and asthma. It is more popular than comparable drugs. There is currently no generic alternative to Dupixent.
Is bamlanivimab FDA approved?
Bamlanivimab is an investigational monoclonal antibody therapy authorized for emergency use by the FDA to treat COVID-19. It is not yet FDA approved, and the safety and effectiveness of this therapy is still being evaluated.
Is Imdevimab a monoclonal antibody?
Drug class: Monoclonal Antibodies. Imdevimab is an investigational monoclonal antibody therapy authorized for emergency use by the FDA to treat COVID-19. It must be administered together with casirivimanb. It is not yet FDA approved, and the safety and effectiveness of this therapy is still being evaluated.
COVID-19 VEKLURYTM (remdesivir)
Following the recent statement from the National Institutes of Health (NIH) COVID-19 Treatment Guidelines Panel about therapies for the COVID-19 Omicron variant, CMS created HCPCS code J0248 for VEKLURY™ (remdesivir) antiviral medication when administered in an outpatient setting.
COVID-19 Monoclonal Antibody Products
The FDA authorized the following investigational monoclonal antibody product under EUA for pre-exposure prophylaxis of COVID-19:
Important Update about Viral Variants
On April 16, 2021, the FDA revoked the EUA for bamlanivimab, when administered alone , due to a sustained increase in COVID-19 viral variants in the U.S. that are resistant to the solo product.
Medicare Coverage for COVID-19 Monoclonal Antibody Products
During the COVID-19 public health emergency (PHE), Medicare will cover and pay for these infusions (when furnished consistent with their respective EUAs) the same way it covers and pays for COVID-19 vaccines.
Coding for the Administration of COVID-19 Monoclonal Antibody Products
CMS identified specific code (s) for each COVID-19 monoclonal antibody product and specific administration code (s) for Medicare payment:
Medicare Payment for Administering COVID-19 Monoclonal Antibody Products
To ensure immediate access during the COVID-19 PHE, Medicare covers and pays for these infusions and injections in accordance with Section 3713 of the Coronavirus Aid, Relief, and Economic Security Act (CARES Act) .
Billing for Administering COVID-19 Monoclonal Antibody Products
Health care providers can bill on a single claim for administering COVID-19 monoclonal antibody products, or submit claims on a roster bill.
